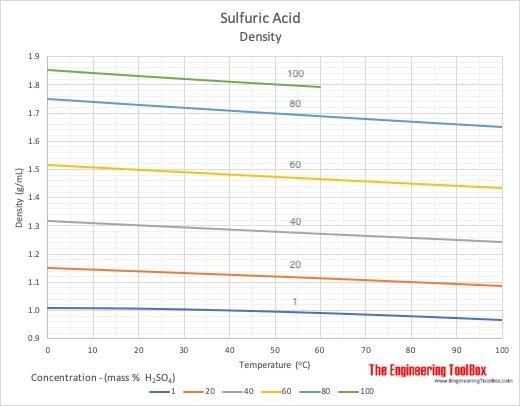
SOLVED: A chemist needs 33.0 g of concentrated sulfuric acid for an experiment. The density of concentrated sulfuric acid at room temperature is 1.84 g/mL. What volume of the acid is required? (
Sulphuric acid, 1 l | Reagents for the Determination of Nitrogen acc. to Kjeldahl | Determination of Nitrogen according to Kjeldahl | Analysis of Food | Inorganic & Analytical Reagents | Chemicals | Carl Roth - International