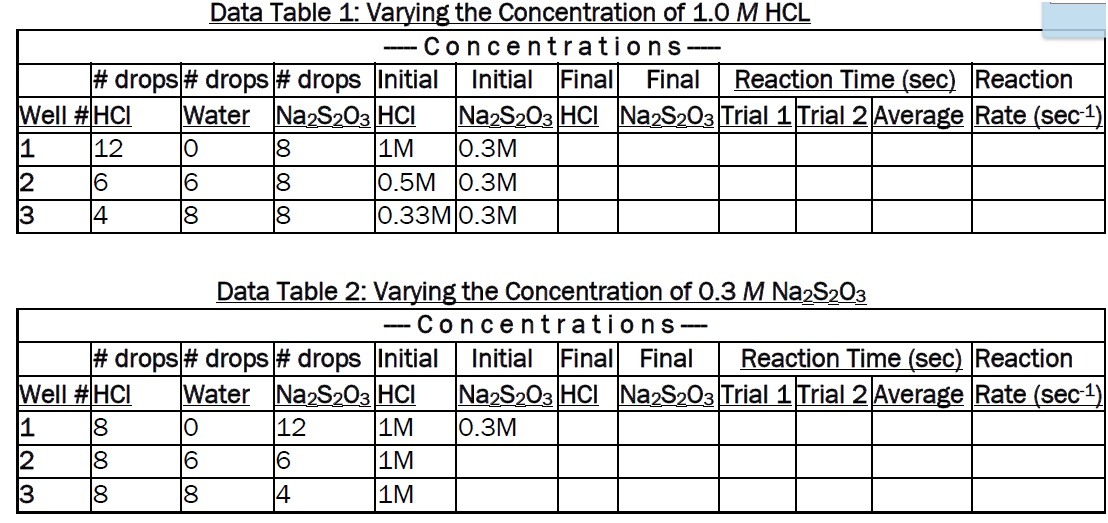
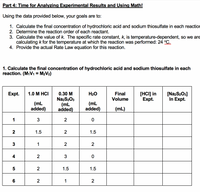
DOC) SCH 4U0 Lab -Rate of Reaction of Sodium Thiosulphate and Hydrochloric Acid | abivarman arul - Academia.edu
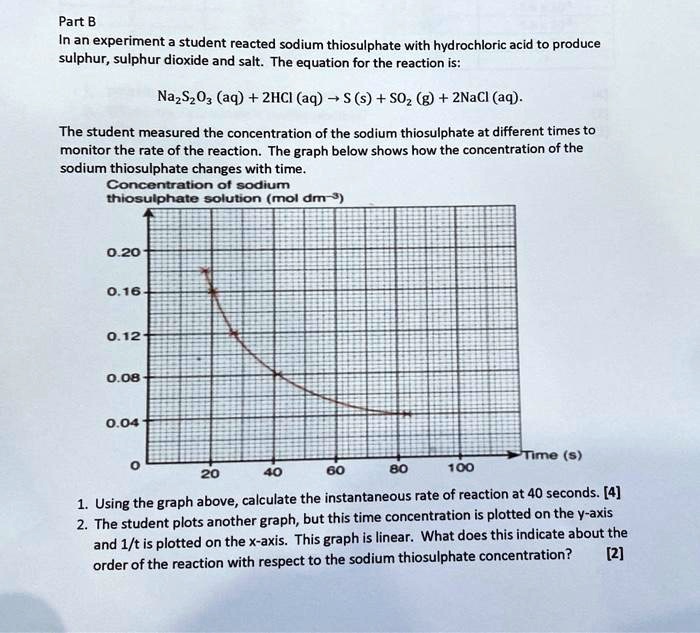
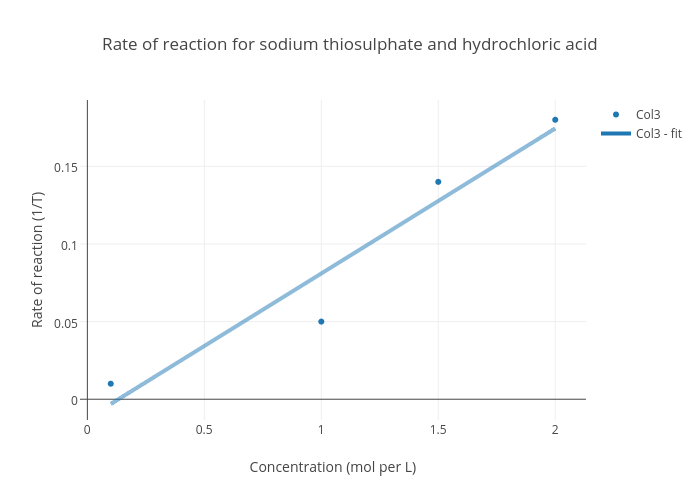
SOLVED: Part B In an experiment student reacted sodium thiosulphate with hydrochloric acid to produce sulphur, sulphur dioxide and salt: The equation for the reaction Is: NazSzO3 (aq) + ZHCI (aq) (s)

To investigate the rate of the reaction between different concentrations of Hydrochloric acid and sodium thiosulphate - GCSE Science - Marked by Teachers.com
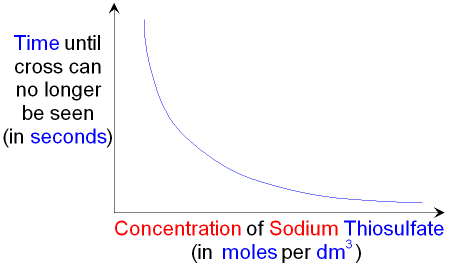
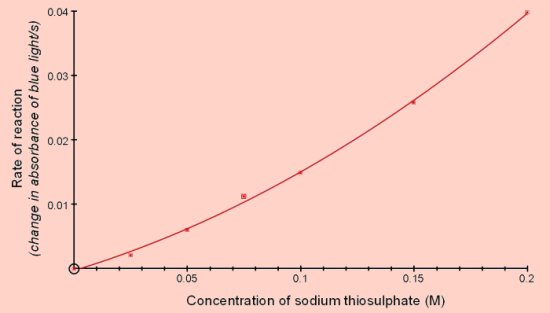
GCSE CHEMISTRY - What is the Effect of Increasing the Concentration on the Rate of a Chemical Reaction? - Collision Theory - GCSE SCIENCE.

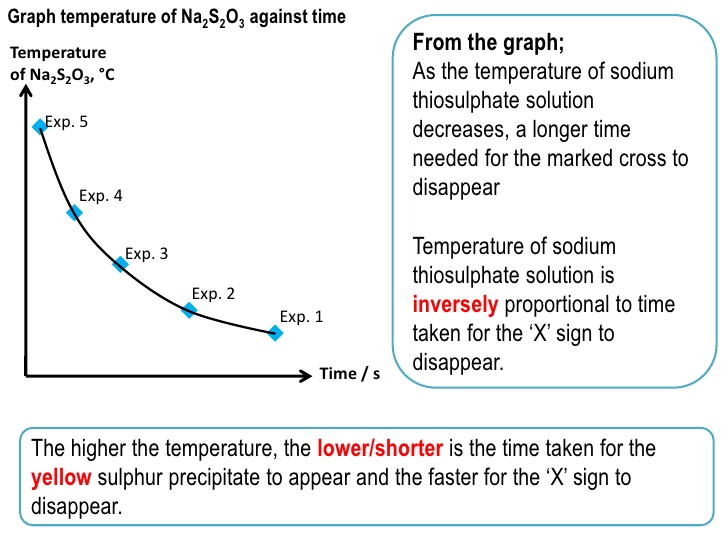
Thiosulphate acid - Determination of the rate of reaction using Sodium Thiosulfate and Hydrochloric - Studocu