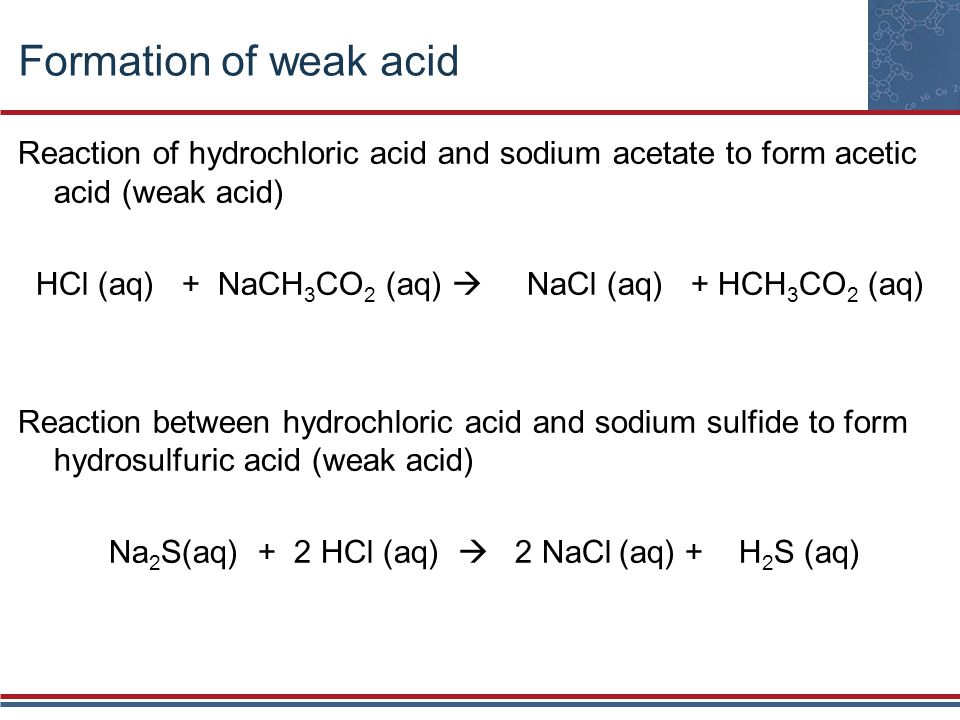
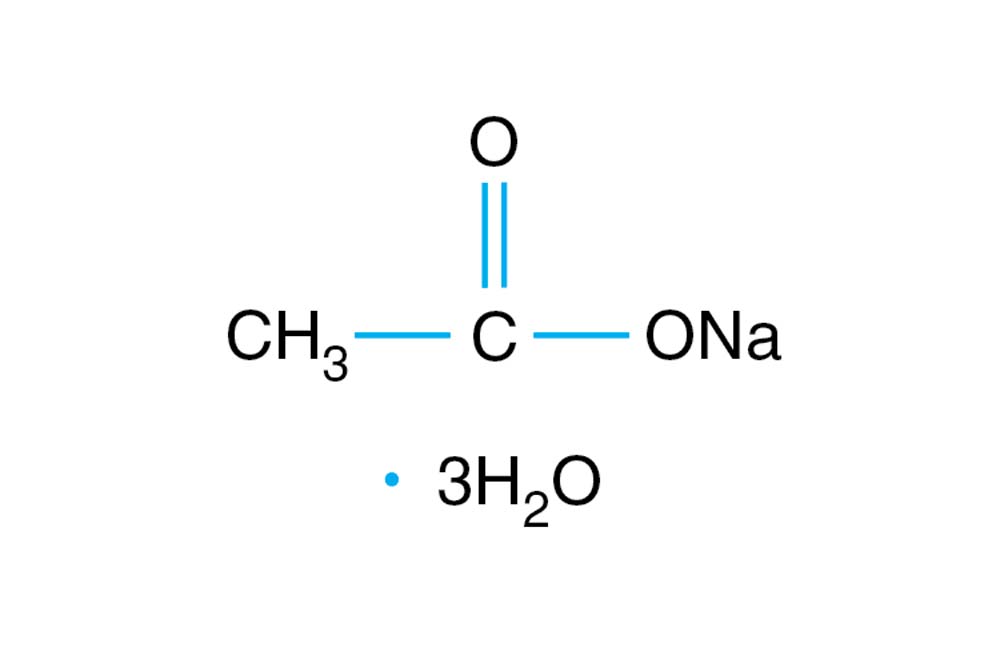
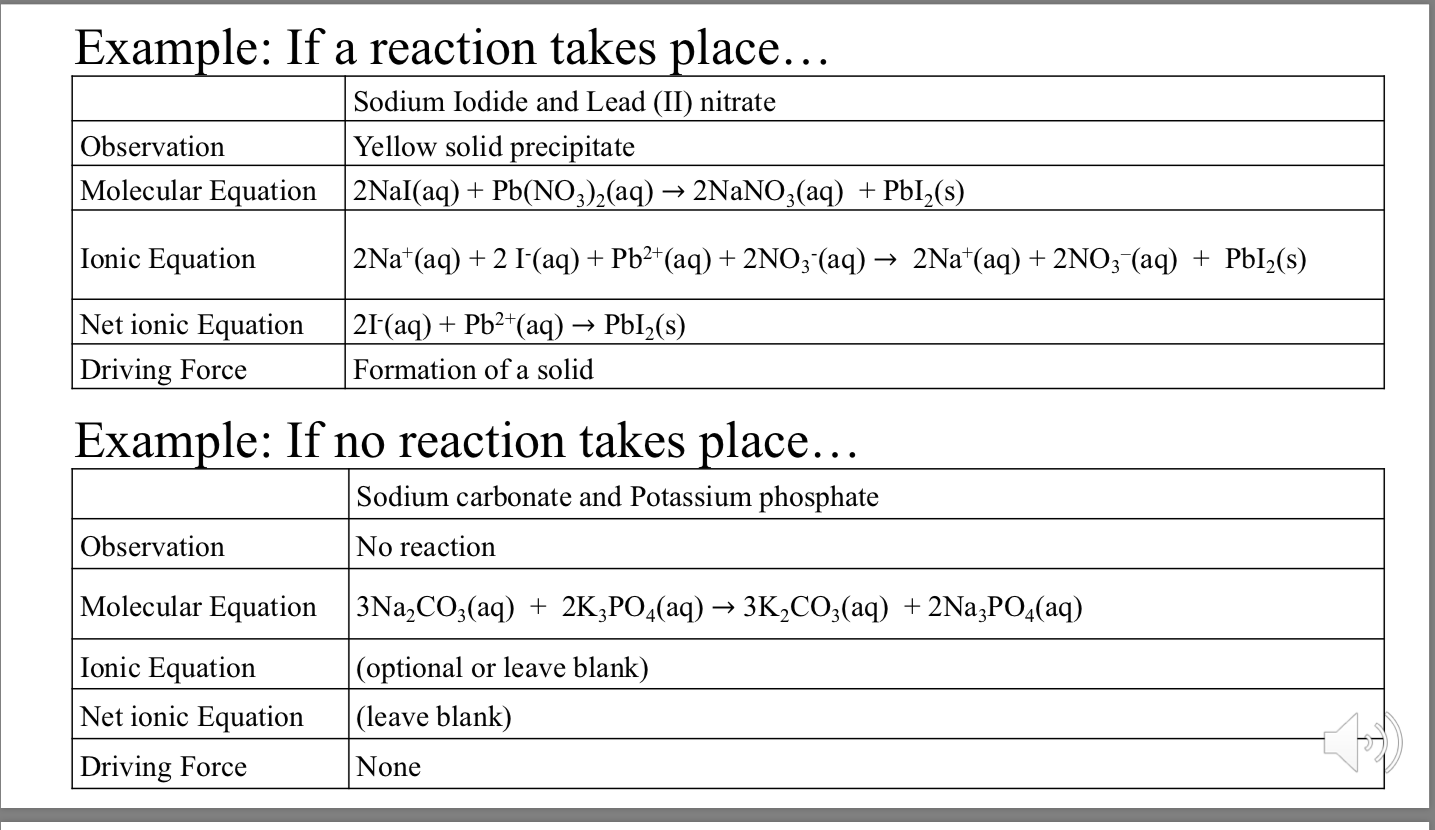
✓ Solved: A quantity of 0.15 M hydrochloric acid is added to a solution containing 0.10 mol of sodium...

20 ml of 0.1 M acetic acid is mixed with 10 ml of sodium acetate. pKa of CH3COOH is 4.8 , then the concentration of sodium acetate, if pH of the mixture is 4.8 is:

SOLVED: buffer solution is made from acetic acid, HCzH;Oz, and sodium acetate, NaCzH;Oz: Suppose small amount of hydrochloric What is the net ionic equation for the reaction that occurs when acid is
When a small amount of HCL is added to a buffer solution of acetic acid and sodium acetate what happen?

SOLVED: What is the net ionic equation for: hydrochloric acid sodium acetate 0 Htlaq) + CH3Oz (aq) HCH;Ozlaq) 0 Ht(aq) + Na (aq)| #HNals) 9 NRI 1D CHlaqk €,HLOH (aq) HNR" CIC

OneClass: If we have a 25.00ml solution containing .038M sodium acetate, NaCH3COO, how much .098M HCl...
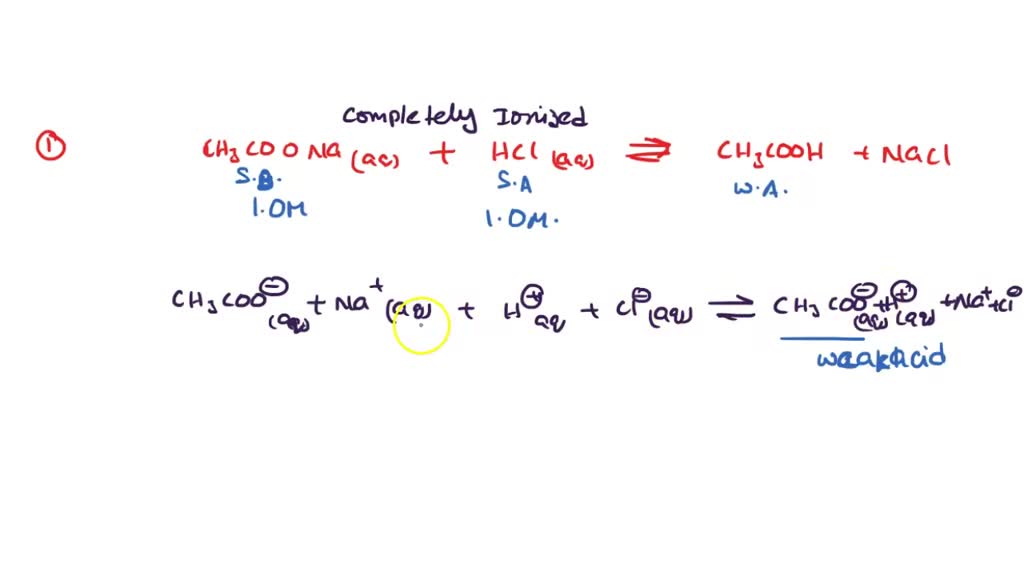
SOLVED: 14) Equal volumes of 1.0 M sodium acetate, NaCH;COz, and 1.0 M hydrochloric acid. HCL are combined. Write the net ionic equation for the reaction and identify the aqueous species that
Compare 1 L of acetate buffer solution (0.50 mol of acetic acid and 0.50 mol sodium acetate) to 1 L of HCl solution Similarities






![Sodium Acetate [CH3COONa] Molecular Weight Calculation - Laboratory Notes Sodium Acetate [CH3COONa] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/sodium-acetate-molecular-weight-calculation.jpg)