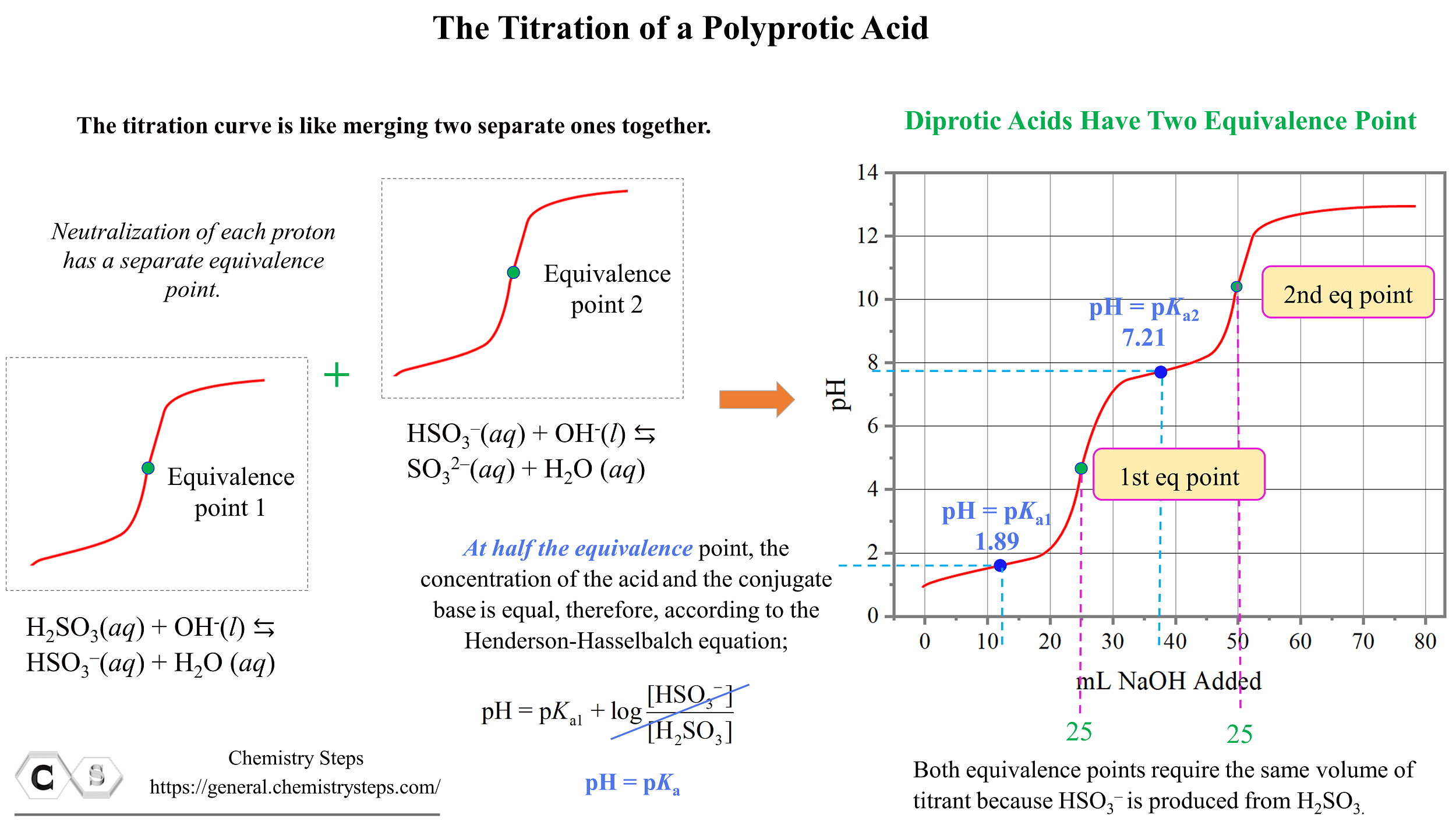
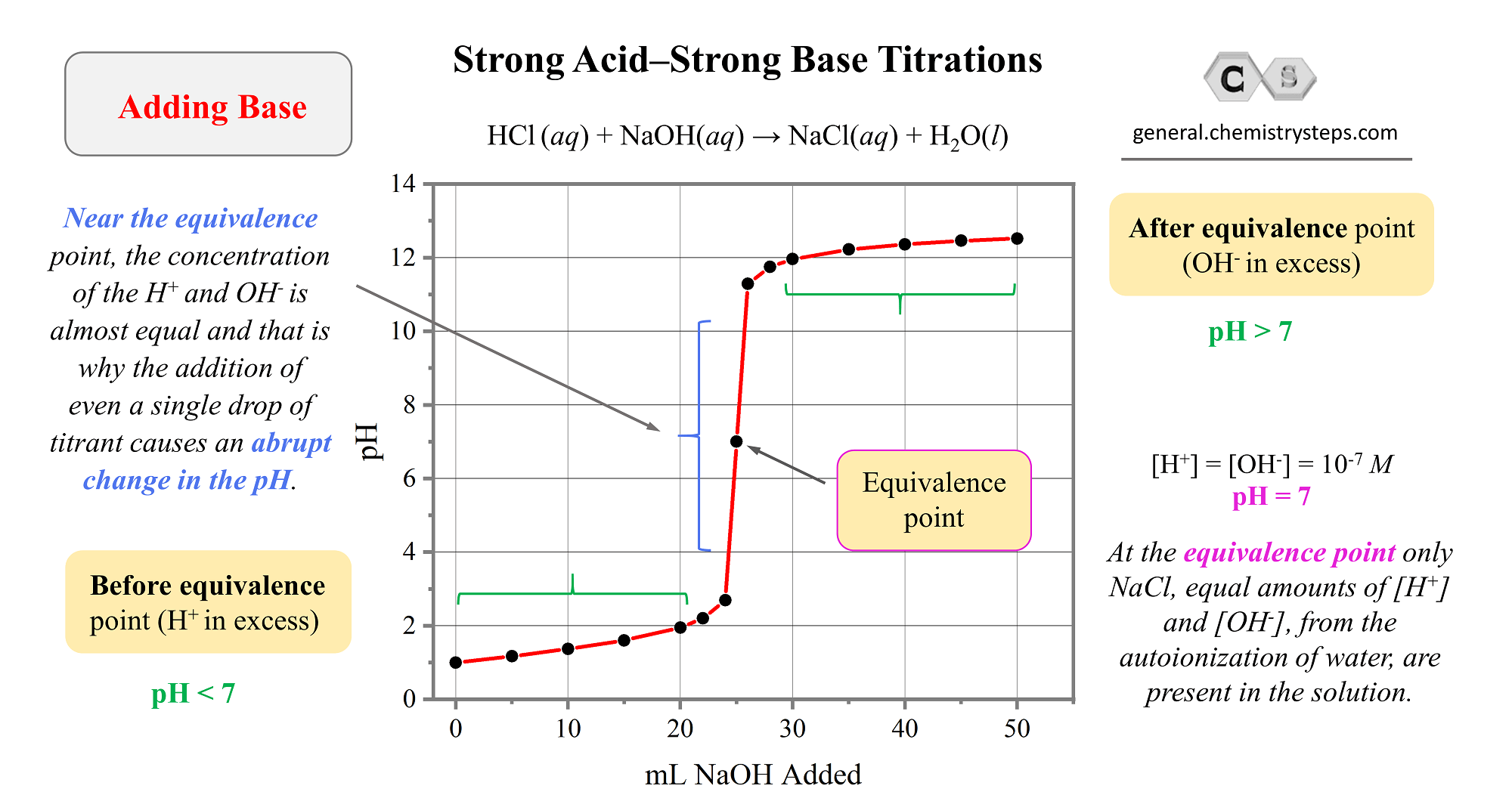
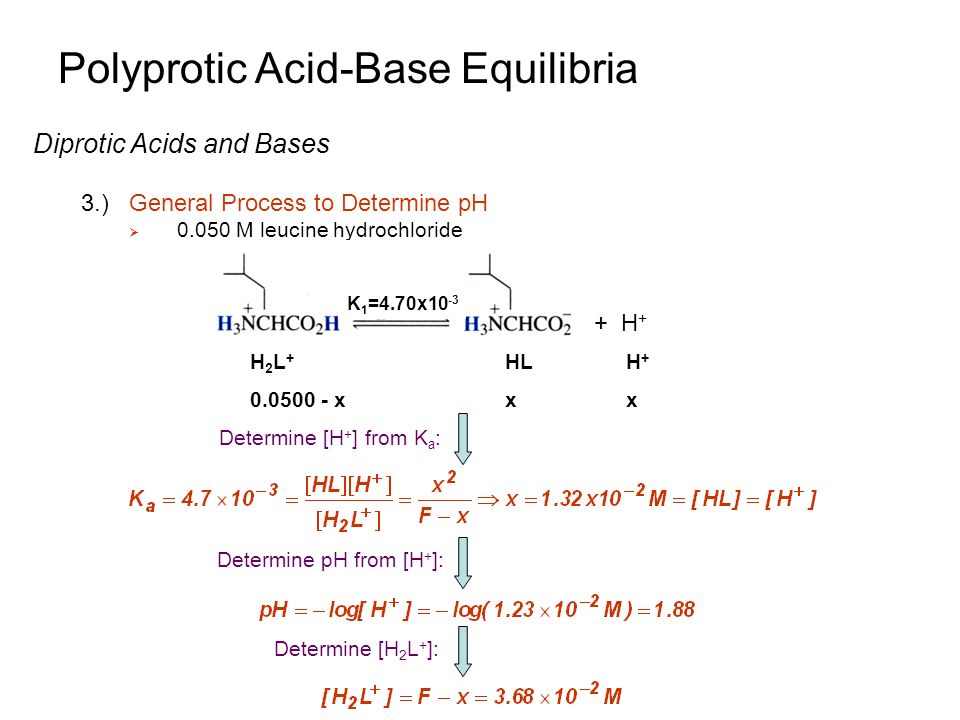
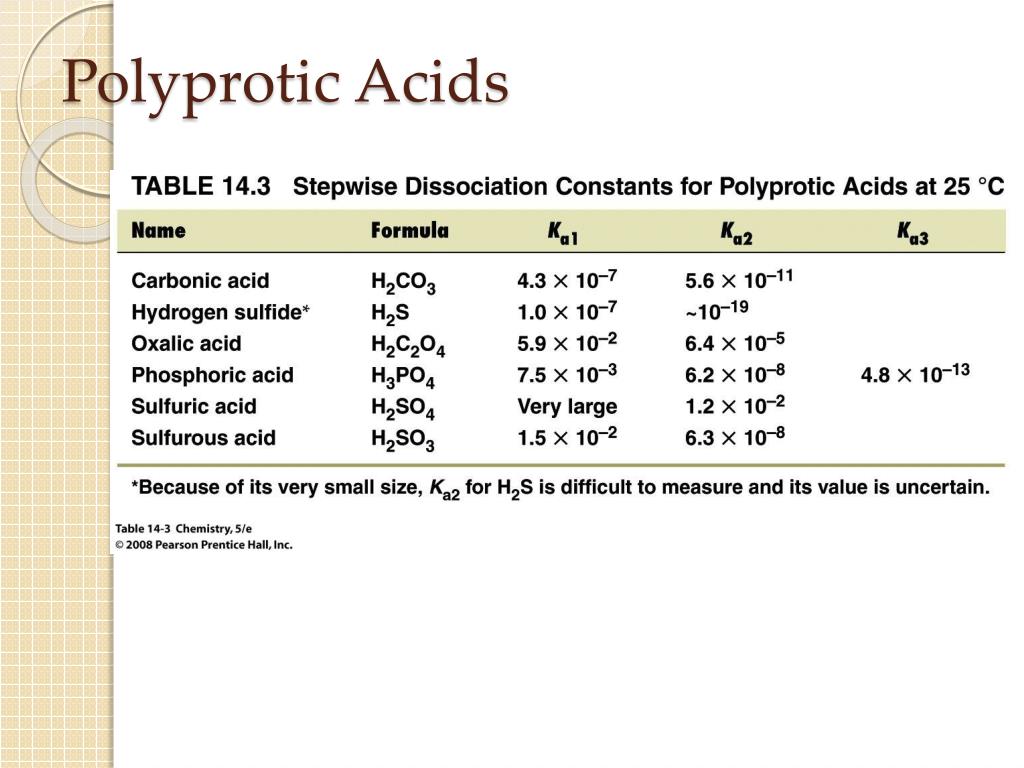
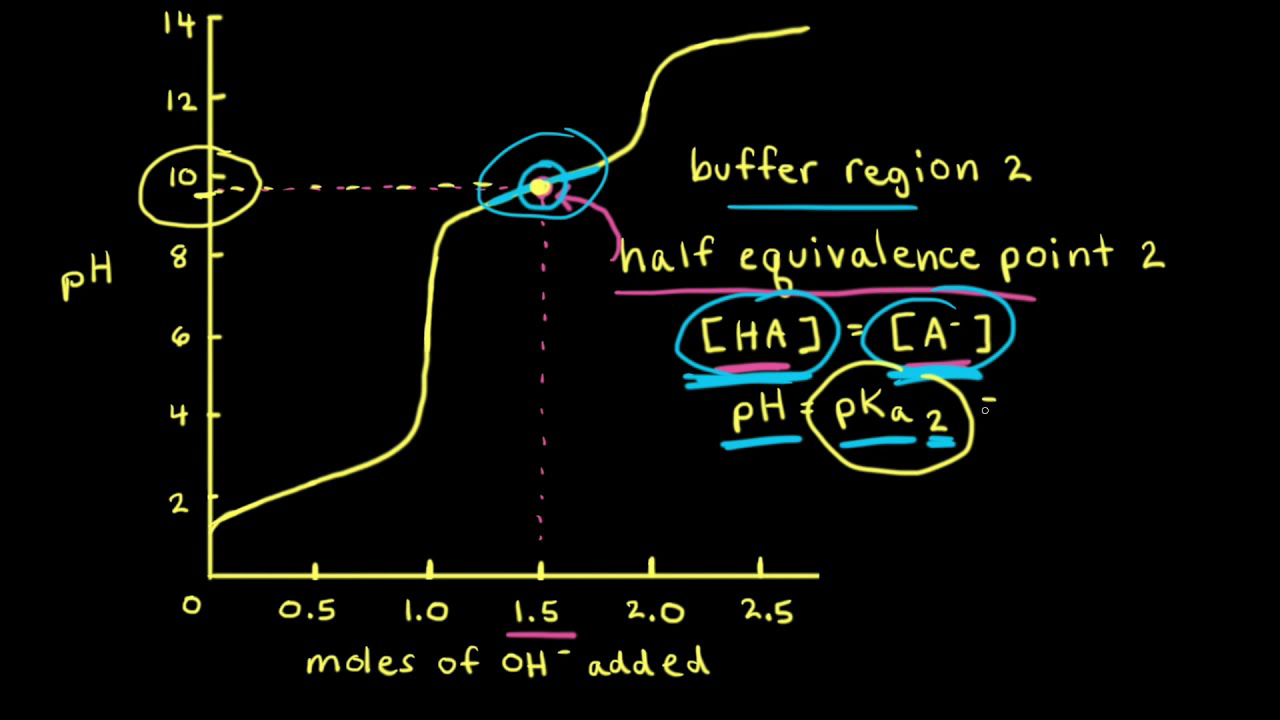
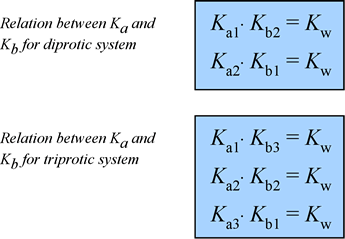
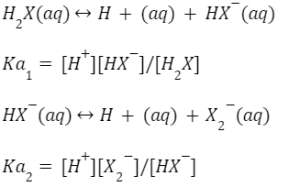For a polybasic acid, the dissociation constants have different values for each step, e.g., H3A H^+ + H2A^- ; K = Ka1 H2A^- H^+ + HA^2 - ; K = Ka2 HA^2 -
Synthesis, characterization and application of a novel polyazo dye as a universal acid–base indicator - RSC Advances (RSC Publishing) DOI:10.1039/D2RA04930A

Di and Polybasic Acids and Bases - Definition, Ionisation, Basicity, Acidity, Examples,with videos , FAQs of Di and polybasic acids and bases.

A Quercetin-Derived Polybasic Acid Hardener for Reprocessable and Degradable Epoxy Resins Based on Transesterification | ACS Applied Polymer Materials

Polyprotic Acid Base Equilibria Problems, pH Calculations Given Ka1, Ka2 & Ka3 - Ice Tables - YouTube

Polybasic Acids - O Level Chemistry & IP Chemistry Notes by 10 Year Series Author - Chemistry Specialist