
molar mass of sulfuric acid/molecular mass/molecular weight of sulphuric acid/ H2SO4 molar mass. - YouTube
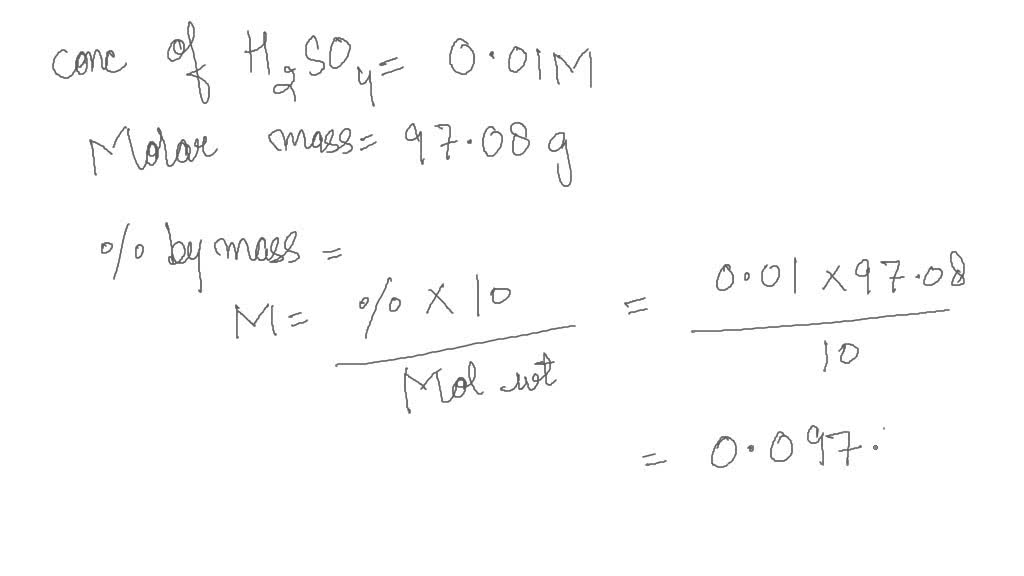
SOLVED: The molarity of a sulfuric acid solution is 0.01 M. Express this concentration in % by mass, ppm, and ppb.(Density of water is 1 g/mL; molar mass of sulfuric acid is

A molecular mass of sulphuric acid is 98 If 49 g of acid is dissolve in water to make - Chemistry - Coordination Compounds - 13542845 | Meritnation.com

Calculate the mass of 1 5 grams molecules of sulphuric acid - Chemistry - Some Basic Concepts of Chemistry - 15496931 | Meritnation.com

14.7 g of sulphuric acid was needed to dissolve 16.8 g of metal. Calculate the equivalent weight of the metal and the volume of hydrogen liberate at NTP .

Calculate molecular mass of the following molecules: a. Sulphuric acid (H(2) SO(4)) b. Glucose (C(6) H(12) O(6)) c. Methane (CH(4))

Calculate the mass percent `(w//w)` of sulphuric acid in a solution prepared by dissovles 4 g of - YouTube

Sulphuric acid and orthophosphoric acid have the same molecular mass. Ratio of the masses of these acids needed to neutralise the same amount of an alakli if the sulphate and dihydrogen orthophosphate
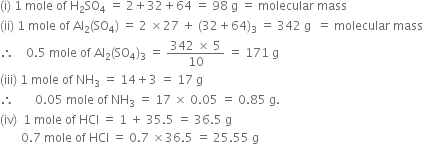
Calculate the mass in grams of the following:(i) One mole of sulphuric acid (H2SO4).(ii) 0.5 mole of aluminium sulphate Al2(SO4)3.(iii) 0.05 mole of ammonia (NH3).(iv) 0.7 mole of hydrogen chloride (HCl). from

The density (in g mL^(-1)) of a 3.60M sulphuric acid solution that is 29% H(2)SO(4) (Molar mass = 98 g mol^(-1)) by mass will be

SOLVED: Q2 m3 A 0.5-molar aqueous solution of sulphuric acid flows into process unit at rate of 1.25 min: The specific gravity of the solution is 1.03 and the molar mass of
![Calculate the mass percentage composition of the elements in sulphuricr acid. [(H = 1, O = 16, S = 32)Atomic mass] Calculate the mass percentage composition of the elements in sulphuricr acid. [(H = 1, O = 16, S = 32)Atomic mass]](https://dwes9vv9u0550.cloudfront.net/images/1829042/c6d30049-457a-4243-bd97-a1c6b7154bcd.jpg)









