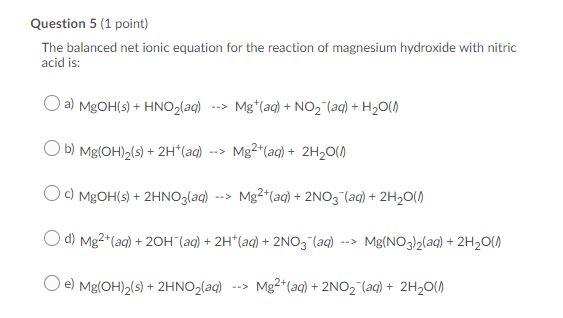SOLVED: 8. Write the correct formulas and balance Classify the reaction as either singie replacement or double replacement (neutralization if an acidlbase reaction) . (16 pts) magnesium nitric acid magnesium nitrate hydrogen

SOLVED: Choose the correct and balanced equation. HNOz MgC (OH) 2 MgHNO: OH- 2 HNO3 Mg(OH)2 Mg(NO3) + 2Hz0 HNO3 + 2 Mg(OH) 2 Mgz NO3 + HzO HNO3 Mg(OH) 2 MgNOz +

Acids and Metals Acids react with certain metals to produce hydrogen gas and the metal salt. metal acid metal salt. - ppt download

Ionic equations A chemical equation shows the number of atoms and molecules of the reactants and products. Also shows physical state of reactants and products. - ppt download
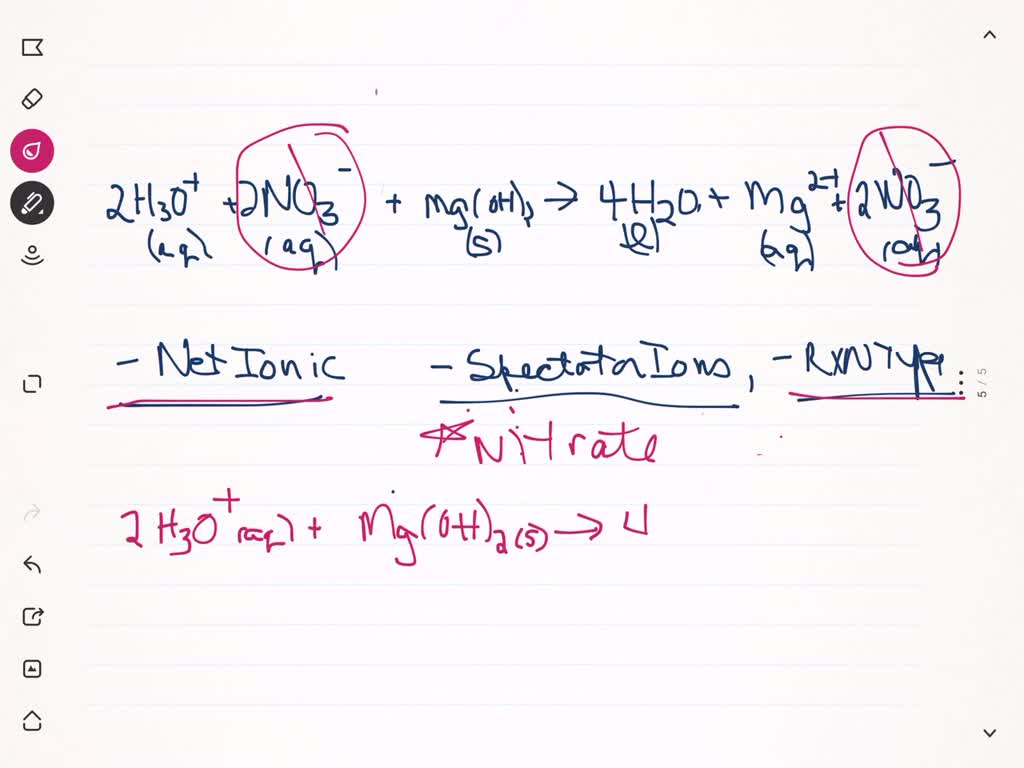
SOLVED:Write the net ionic equation and identify the spectator ion or ions in the reaction of nitric acid and magnesium hydroxide. What type of reaction is this? 2 H3O^+(aq)+2 NO3^-(aq)+Mg(OH)2(s) → 4

SOLVED: Answer question 9a and 9b using the reaction equation between nitric acid reacts with magnesium hydroxide. 2 HNO; (aq) MglOH)z (aq) + Mg(NOs)z (aq) 2 HzO (aql 9a.A 35.00 mL of

Mg(OH)2+HNO3=Mg(NO3)2+H2O Balanced Equation||Magnesium Hydroxide+Nitric acid =Magnesium Nitrate+Water - YouTube


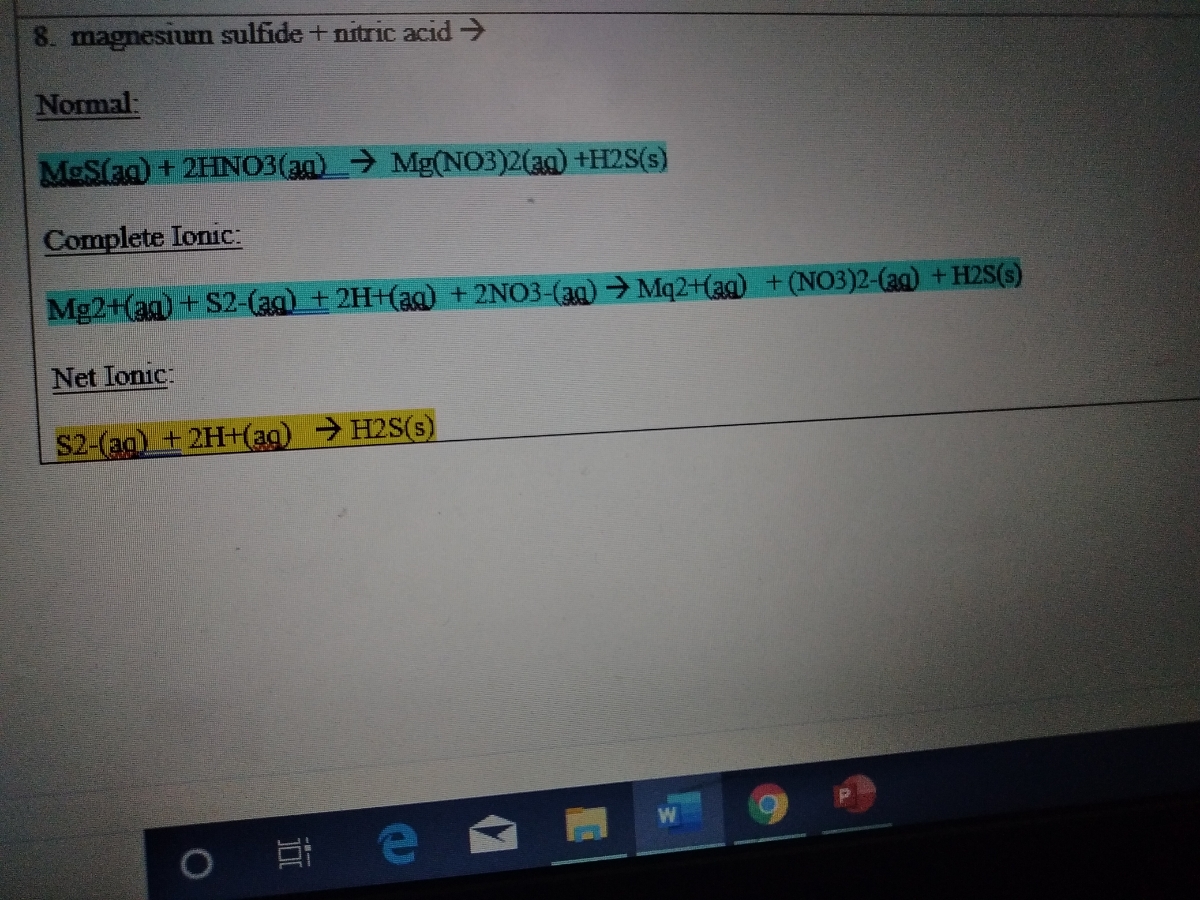
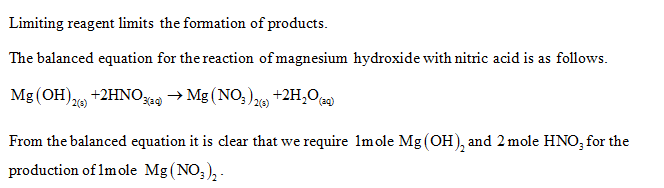






.PNG)