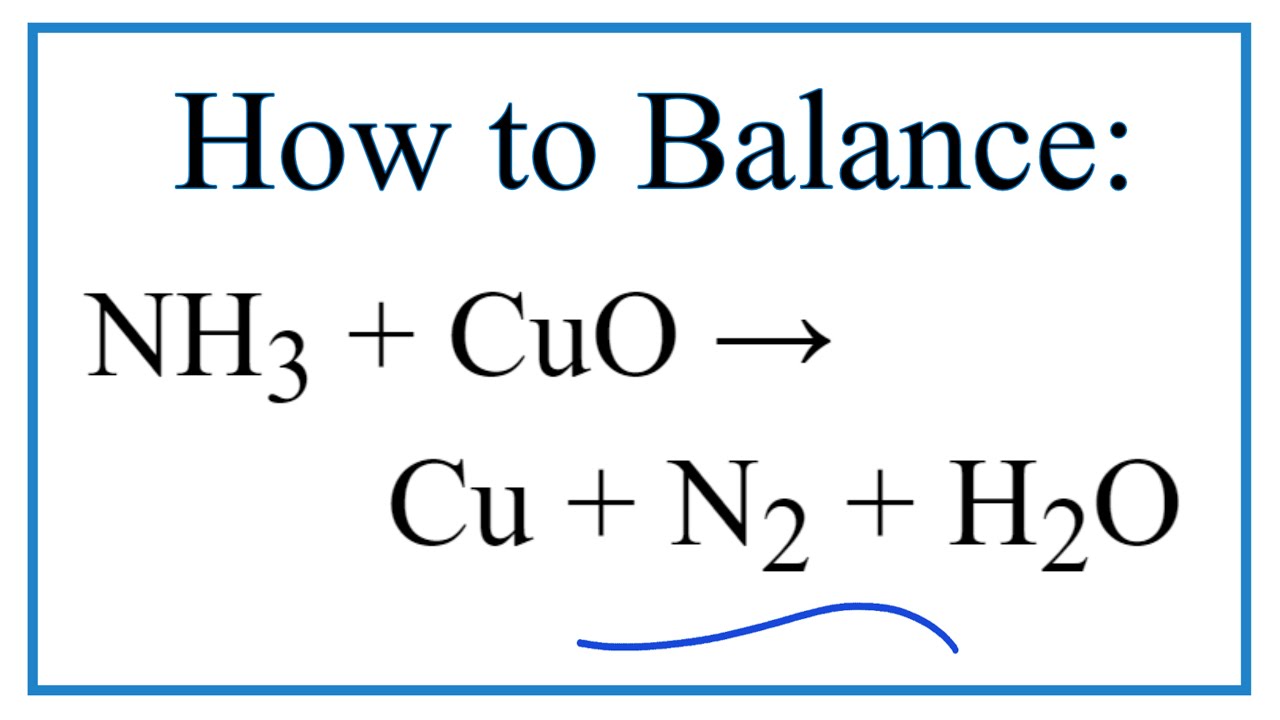
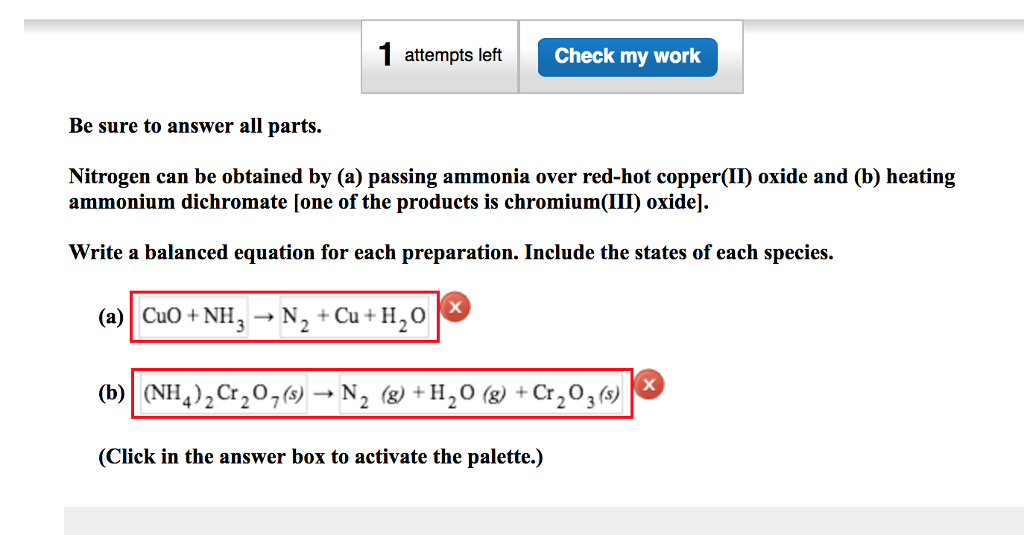
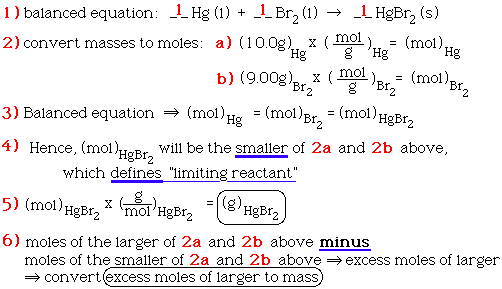Give balanced equation for each of the following:Reduction of hot Copper (II) oxide to copper using ammonia gas:

Electrocatalytic, Homogeneous Ammonia Oxidation in Water to Nitrate and Nitrite with a Copper Complex | Journal of the American Chemical Society

When ammonia is passed over heated copper oxide, the metallic copper is obtained, the reaction shows - YouTube
Dry ammonia gas was passed over heated lead (II) oxide and the products passed over anhydrous copper - Tutorke

Coatings | Free Full-Text | Electro-Oxidation of Ammonia over Copper Oxide Impregnated γ-Al2O3 Nanocatalysts
When dry ammonia gas is passed over hot copper (II) Oxide, a shinny brown residue and a colourless droplets are formed. Explain these two observations

When ammonia is passed over heated copper oxide, the metallic coper is obtained. The reaction shows - YouTube

Copper(II) oxide reacts with ammonia to give copper, water and nitrogen. CuO((s)) +NH(3(g)) to xCu((s))+yH(2)O((l)) +zN(2(g)) The value of (x + y + z) is

Copper based catalysts for the selective ammonia oxidation into nitrogen and water vapour—Recent trends and open challenges - ScienceDirect

When ammonia is passed over heated copper oxide, the metallic copper is obtained, the reaction shows - YouTube
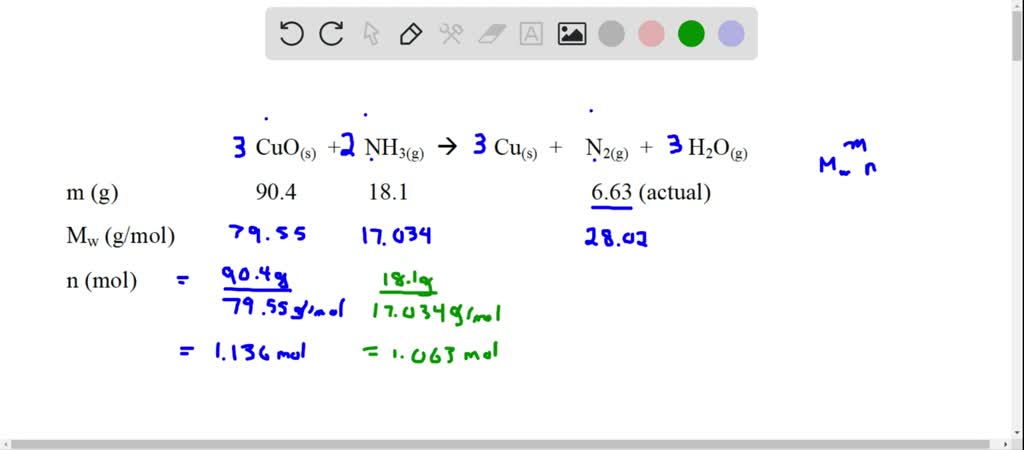
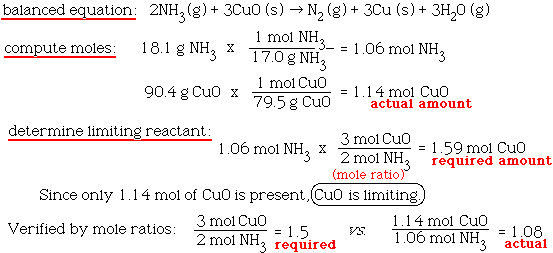
SOLVED: Gaseous ammonia, NH3, and copper(II) oxide, CuO, react together to form gaseous nitrogen (N2), solid copper, and water vapor. If a sample containing 17.9 g of NH3is reacted with 90.0 g

Coatings | Free Full-Text | Electro-Oxidation of Ammonia over Copper Oxide Impregnated γ-Al2O3 Nanocatalysts

Selective catalytic oxidation of ammonia to nitric oxide via chemical looping | Nature Communications
![State one obervation for the following : Ammonia gas is passed over heated copper [II] oxide. | ... - YouTube State one obervation for the following : Ammonia gas is passed over heated copper [II] oxide. | ... - YouTube](https://i.ytimg.com/vi/7deWf3VEtQk/maxresdefault.jpg)