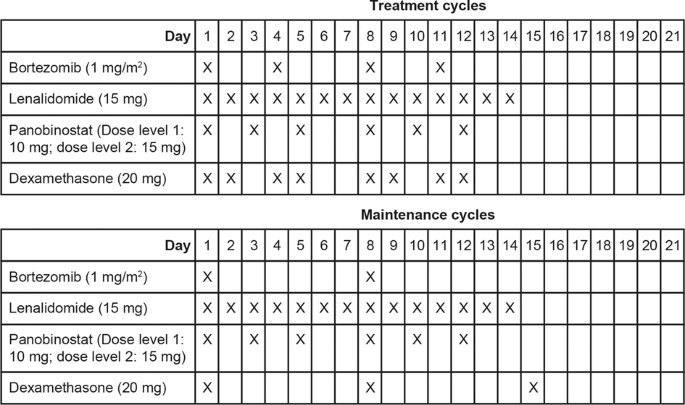
RIASSUNTO DELLE CARATTERISTICHE DEL PRODOTTO 1. DENOMINAZIONE DEL MEDICINALE Bortezomib Teva 2,5 mg polvere per soluzione iniett

Teva Announces Launch of a First Generic Version of Revlimid® (lenalidomide capsules), in the U.S. | Business Wire